Dwarf Mistletoe Rating

The severity of dwarf mistletoe infection (number of infections relative to tree size) is universally measured by the Hawkworth 6-class dwarf mistletoe rating (DMR) system [5]. It is used in discussing ecology and management of these diseases, so let’s get familiar with it first. It’s pretty simple:
- Divide the live crown into thirds. If you are too close to the tree, there is a tendency to make the lower third too small and the upper third too large.
- Rate the severity of each third on a scale of 0-2:
- 0 – no infection
- 1 – half or fewer of primary branches are infected
- 2 – over half of primary branches infected
- Sum the thirds to get tree DMR.
- If there are stem but no branch infections, give the tree a DMR of 1.
- For stand DMR, average the DMR of all host trees (including uninfected).
Lodgepole pine reaches an average DMR of 5 within about 70 years after 1st infection [6].
Increases in mistletoe spread and severity due to human influence
In presettlement forests of western USA, it is generally considered that dwarf mistletoes tended to be patchily distributed [9]. Some early management practices tended to increase the abundance and distribution of dwarf mistletoes, and some (especially fire exclusion) continue to do so (see Stand History and Fire sections below). Thinning commonly practiced in ponderosa pine of the Southwest (“improvement selection”) may have prevented mortality due to mistletoe, but also contributed to its spread and intensification [1, 4, 7]. Selectively harvesting the most valuable trees (high-grading) concentrated the mistletoe in the overstory while creating opportunities for reproduction, establishing ideal conditions for dwarf mistletoe spread and intensification. Similarly, incomplete clearcuts left unmerchantable, infected trees that led to heavy infection of the regeneration. Excessive grazing, road building, and direct fire suppression have decreased fire frequency, enhancing multi-story, dense stands that are more susceptible to dwarf mistletoes in some forest types, while at the same time removing the single most important natural control of dwarf mistletoes.
These factors contributed to an increase in the distribution and abundance of dwarf mistletoes in many forests of the western United States. In ponderosa pine forests of the Southwest, for instance, it is widely acknowledged that past overgrazing, fire suppression and logging practices have resulted in increased distribution and severity of southwestern dwarf mistletoe [13]. Surveys conducted in the 1950s and 1980s indicate increase in the distribution of southwestern dwarf mistletoe on National Forests of Arizona and New Mexico from 30% to 38% of the ponderosa pine type, a 27% increase [10]. Similar surveys over a 41-year period on the Bighorn National Forest indicate continuing increase in incidence of lodgepole pine dwarf mistletoe from 31% in 1958, to 36% in 1978, and a conservative estimate of 44% in 1999 [3], an increase of 42% in only 41 years. Forest changes since European settlement east of the Cascades in Washington and Oregon are associated with increased distribution and severity of dwarf mistletoes in Douglas-fir and true fir forests [8]. Because of changes in fire frequency, a similar increase in western dwarf mistletoe is inferred for ponderosa pine forests in that region. Douglas-fir dwarf mistletoe has increased in the Inland West where historic practices have made the host more widespread and continuous [2].
Logging and changes in fire regime likely increased mistletoe infection over large areas in lodgepole pine forests of Colorado. Dwarf mistletoe infection in lodgepole pine increased with time since the last fire, or decreased as fire frequency increased, in several studies, and a general increase in dwarf mistletoe intensity and distribution over time was anticipated due to changes in fire return interval [9, 15]. In lodgepole pine forests of eastern Oregon and Washington, although dwarf mistletoe was undoubtedly severe in some presettlement forests, it is now more widely distributed and carrying over between stands now partially replaced by mountain pine beetle rather than more completely replaced by fire [8]. In lodgepole pine of western Montana and northern Idaho, the increase in dwarf mistletoe, together with fire exclusion and increasing mountain pine beetle vulnerability, have created a huge potential for fires [11]. An exception is the Targhee National Forest in Idaho, where incidence of dwarf mistletoes declined in both lodgepole pine and Douglas-fir between 1978 and 1996 [14]. In that case, widespread bark beetle outbreaks and threat of wildfire led to a huge effort at salvage, regeneration and seedling protection that shifted forest structure from mostly mature, heavily infested stands to younger, lightly infested stands.
Stand structure
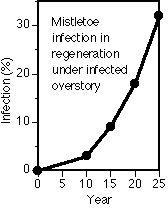

Spread and intensification is much faster from overstory to understory than in an even-aged stand. At left is percent of trees infected over time in an understory with an infected overstory.
Similarly, reproduction adjacent to an infected stand is infected faster than an even-aged stand. At right, you can see that infection tails off as distance from the residual stand increases.
Host vigor
There is a generalization that obligate parasites do better on vigorous than on stressed hosts. With mistletoes, it isn’t that simple. Indeed, the parasite grows more vigorously on a vigorous host, producing more pollen or seeds, but other features of a vigorous host decrease success of the mistletoes. Think about an infection on the lower crown. It needs to have its progeny climb the tree to be successful. But a vigorous tree has a fuller crown with greater retention of needles. Now on the one hand this increases the chance of catching seeds, but on the other hand seeds will have a hard time shooting very high. Just as important, the live crown will be climbing faster than the mistetoe may be able to keep up. If the tree grows in height significantly faster than the mistletoe climbs, infection will never reach the upper or middle crown and may even be lost [12].
Stand density

Tree density can have a strong influence on spread rate (horizontal distance through the stand over time). Clearly the spread rate will be 0 when tree spacing exceeds the maximum dispersal distance (roughly 16 m or 52 ft). As density increases, more seeds will successfully make the jump and spread rate increases. However, as density increases further, seeds are intercepted before they get very far, so spread rate decreases again [4].
The graph shows relative rate of dwarf mistletoe spread in relation to tree spacing for ponderosa pine 30-40 years old in the Southwest (based on data from [4]). The curve would be shifted to left for younger stands and to the right for older stands. As a practical matter, within the range of densities usually found in managed forests (on the left side of the graph), mistletoes spread more rapidly in open than in dense stands [12].
Stand composition
Host specificity obviously restricts the ability of mistletoes to spread and intensify in mixed stands.
Stand history

Logging influences many of the factors already discussed that in turn influence mistletoes. Past cutting practices that did not heed the long-known methods of mistletoe control created many of today’s problems. High grading and selective logging created open, multistoried stands with reduced species complexity. The worst situation is heavy cutting that left the worthless, heavily infected trees from the previous trees standing to infect the regeneration. That was fairly common in the old days. On the other hand, extensive clearcutting in other areas, where complete, has eradicated mistletoes from large areas. Fire history has its own section.
Fire
Although its effects can vary, fire is generally considered the most important natural control of dwarf mistletoes. Where fire is intense and widespread enough to kill all trees in large patches, it effectively eradicates the pathogen. Generally trees will recolonize such burned areas much faster than mistletoe, especially fire-adapted species like lodgepole pine. There are extensive areas of lodgepole that today are relatively free of mistletoe because of extensive and intense fires in the past century. Even with low-intensity ground fires, fire tends to torch and essentially sanitize mistletoe infection centers because of their fuel conditions: dead standing and downed trees, brooms with concentrated foliage, resin and trapped needles, and resinous branches, functioning as a fire ladder and leading to locally increased fire intensity.
However, it is also possible for fire to have no net sanitizing effect or even the opposite, when it creates opportunities for natural regeneration adjacent to surviving, infected trees.
Long-range dispersal
Birds and other animals can probably accomplish long-range dispersal. Seeds of dwarf mistletoes have been found on such animals, so we have to assume that occasionally such a seed will be deposited on a suitable infection court. The only question is how often this happens and results in establishment. And think of this: you need not just one seed to be successfully dispersed this way, but at least two, male and female, to the same area (if it’s within pollination distance, it’s not really long distance). This may be a factor in migration of the species over hundreds and thousands of years. It is probably not important epidemiologically within the time frame of management. Thus, from a practical standpoint, it can be ignored.
Related pages include:
References
- 1.Conklin DA. 2000. Dwarf Mistletoe Management and Forest Health in the Southwest. USDA Forest Service, Southwestern Region <https://www.fs.usda.gov/Internet/FSE_DOCUMENTS/stelprdb5238460.pdf>.
- 2.Hadfield JS, Mathiasen RL, Hawksworth FG. 2000. Douglas-fir dwarf mistletoe. Forest Insect and Disease Leaflet 54. USDA Forest Service, Washington DC, USA <https://www.fs.usda.gov/Internet/FSE_DOCUMENTS/fsbdev2_043663.pdf>.
- 3.Harris JL. 2003. 1999 road-plot survey of dwarf mistletoe and comandra blister rust diseases of lodgepole pine on the Bighorn National Forest. Biological Evaluation R2-03-07. Golden, Colorado, USA: USDA Forest Service, Rocky Mountain Region, Renewable Resources. 13 pp. <https://forestpathology.org/pdfs/harris_r2-03-07.pdf>.
- 4.Hawksworth FG. 1961. Dwarfmistletoe of ponderosa pine in the Southwest. Technical Bulletin 1246. USDA Forest Service, Rocky Mountain Forest and Range Experiment Station, Washington DC, USA.
- 5.Hawksworth FG. 1977. The 6-Class Dwarf Mistletoe Rating System. General Technical Report RM-48. USDA Forest Service, Rocky Mountain Forest and Range Experiment Station, Fort Collins, Colorado, USA <https://archive.org/details/CAT10837363>.
- 6.Hawksworth FG, Hinds TE. 1964. Effects of dwarf mistletoe on immature lodgepole pine stands in Colorado. Journal of Forestry 62(1):27–32 <https://academic.oup.com/jof/article/62/1/27/4672153>.
- 7.Heidmann LJ. 1983. Silvicultural control of dwarf mistletoe in southwestern ponderosa pine. Research Note RM-433. USDA Forest Service, Rocky Mountain Forest and Range Experiment Station, Fort Collins, Colorado, USA <https://www.google.com/books/edition/Silvicultural_Control_of_Dwarf_Mistletoe/ckYzkwV-dhwC>.
- 8.Hessburg PF, Mitchell RG, Filip GM. 1994. Historical and current roles of insects and pathogens in eastern Oregon and Washington forested landscapes. U.S. Department of Agriculture, Forest Service, Pacific Northwest Research Station <10.2737/pnw-gtr-327>.
- 9.Kipfmueller KF, Baker WL. 1998. Fires and dwarf mistletoe in a Rocky Mountain lodgepole pine ecosystem. Forest Ecology and Management, pp. 77–84 <10.1016/s0378-1127(98)00217-5>.
- 10.Maffei H, Beatty JS. 1988. Changes in the incidence of dwarf mistletoe over 30 years in the Southwest. In: Proceedings 36th Western International Forest Disease Work Conference, Park City, Utah, ed van der Kamp B, pp. 88–90. Western International Forest Disease Work Conference.
- 11.Monnig E, Byler JW. 1992. Forest Health and Ecological Integrity in the Northern Rockies, 2nd edition. FPM Report 92-7 (R1-92-130). USDA Forest Service, Northern Region, Missoula, Montana, USA.
- 12.Parmeter JR. 1978. Forest stand dynamics and ecological factors in relation to dwarf mistletoe spread, impact and control. In: Proceedings of the symposium on dwarf mistletoe control through forest management, April 11-13, 1978, Berkeley, California. General Technical Report PSW-31, eds Scharpf RF, Parmeter JR, pp. 16–30. Berkeley, CA: USDA Forest Service, Pacific Southwest Forest and Range Experiment Station.
- 13.Pollock MM, Suckling K. 1995. An ecologically integrated approach to management of dwarf mistletoe (Arceuthobium) in Southwestern forests. Center for Biological Diversity. <https://www.biologicaldiversity.org/swcbd/PROGRAMS/science/MISTLTOE.HTML>.
- 14.Smith JP, Hoffman JT. 1998. Dwarf mistletoe surveys on the Targhee National Forest. 1978 survey results compared to 1996 conditions. Report R4-98–03. USDA Forest Service, Intermountain Region, Forest Health Protection, Ogden, Utah, USA.
- 15.Zimmerman GT, Laven RD. 1984. Ecological interrelationships of dwarf mistletoe and fire in lodgepole pine forests. In: Biology of dwarf mistletoes: Proceedings of the symposium; 1984 Aug. 8; Fort Collins, CO, pp. 123–131. Fort Collins, CO: USDA Forest Service, Rocky Mountain Forest and Range Experiment Station.

